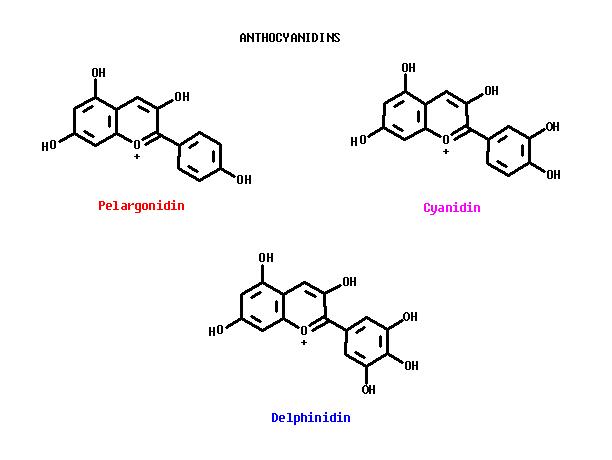

The anthocyanins are common plant pigments. They are found as colored, water-soluble glycosides; with some or all of the sugar groups removed, the colored aglycones are not stable. The color comes from a positive or negative charge distributed over the aryl-chroman ring system. The colors of the charged anthocyanin pigments are dependent on the pH of the intracellular medium containing these pigments.
Anthocyanin pigments in flowers are generally pink, red, or purple. Cyanidin is a red or pink pigment; pelargonidini is the brick-red pigment of geraniums. Both Cyanidin and Pelargonidin are found in Hemrocallis. The blue anthocyanin pigment, Delphinidin, is also found in Hemerocallis, but apparently in quantities too small to produce a blue appearance. Or perhaps the internal pH of the cell vacuole is not high enough to produce the blue color. The biosynthesis proceeds from colorless metabolic intermediates to Chalcone:
Chalcone => Naringenin => Flavonone => dihydro-Kaempferol => dihydro-Quercitin => Leucocyanidin (colorless) => => Anthocyanidin 3-glucoside (colored)
For a more extensive discussion, see FastPlants at the University of Wisconsin. For a metabolic chart of anthocyanin biosynthesis, see HORT640C at Purdue University, and select the links to "Anthocyanin biosynthesis (maize and Arabidopsis genes)" and to "Anthocyanin modifications in Petunia ". There is an interesting paper on conversion of Leucocyanidin to Anthocyanidin 3-glucoside by Nakajima et al. in J. Biol Chem (2001).
Additional links to anthocyanin and delphinidin pigments:
http://antoine.fsu.umd.edu/chem/senese/101/features/water2wine.shtml
http://www.fortunecity.com/roswell/avebury/97/rose.htm