 Shields
Gardens
Shields
Gardens

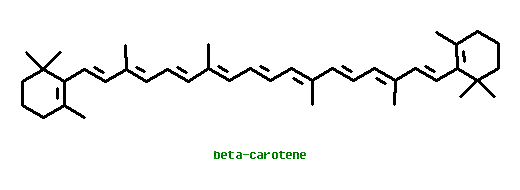
Carotenes are polyunsaturated hydrocarbons formed biochemically from polymerization of isoprendoid compounds. They are yellow, orange, or red, depending on their chemical structures. Carotenoids are soluble in fats, whereas anthocyanins and flavonols are water-soluble compounds because of their attached sugar groups.
Mevalonic acid leads eventually to Geranylgeranyl pyrophosphate (GG-PP), which is the precursor of the first C40 molecule. Two molecules of this compound, GG-PP, are condensed into one molecule of cis-Phytoene, a hydrocarbon with 40 carbon atoms and 9 double bonds.
Phytoene is converted to Lycopene, a C40 polyunsaturated hydrocarbon with 13 double bonds, through stepwise removal of pairs of hydrogen atoms. Many intermediates have been isolated. Lycopene is the red pigment in tomatoes.
Lycopene is converted to beta-Carotene, still a C40 polyunsaturated hydrocarbon, by cyclization to form a cyclohexene ring at each end of the chain.
Beta-Carotene is oxidized on one of the cyclohexene rings to form Zeaxanthin. Zeaxanthin is an example of a Xanthophyll.
Epoxidation of Zeaxanthin on each ring leads to Violaxanthin. Violaxanthin can be converted back to Zeaxanthin in plants.
The overall carotenoid pathway